Australian teenagers will be getting a free vaccine to protect them against four strains of the potentially deadly infection meningococcal.
The combined vaccine for meningococcal strains A, C, W and Y will be available at no cost to Australians aged 14 to 19 from next April.
The change is being listed for the age group on the National Immunisation Program, which provides free jabs.
Meningococcal survivor Lily O’Connell nearly lost her life to the W strain on Christmas Day.
She thought she had the flu, but then a rash appeared on her face and she was rushed to the hospital.
News that makes sense
Your trusted source for staying up-to-date with the world around you. Get free daily news updates and analysis, straight to your inbox.
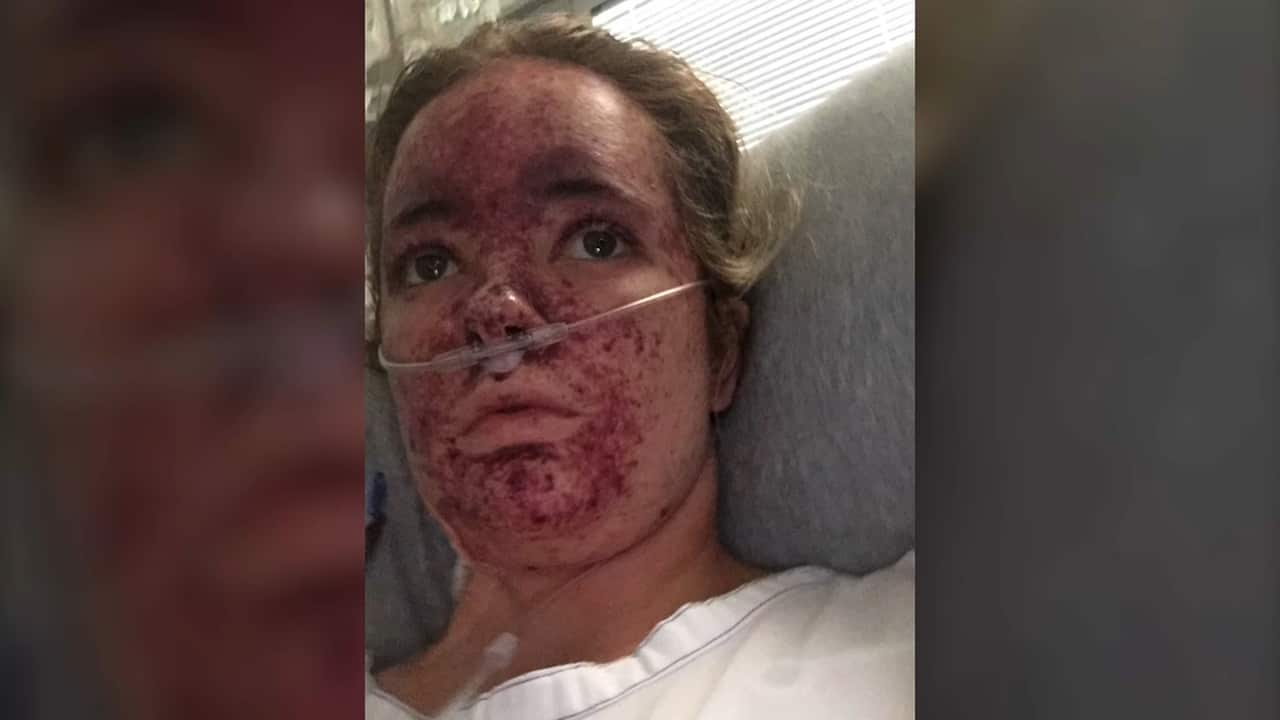
While Ms O’Connell survived, the disease destroyed her kidneys, and she's had to have a transplant.
She said the new national roll-out of vaccines was a crucial step in saving lives.
"It's a great initiative and it's come at a very important time," she told SBS News.
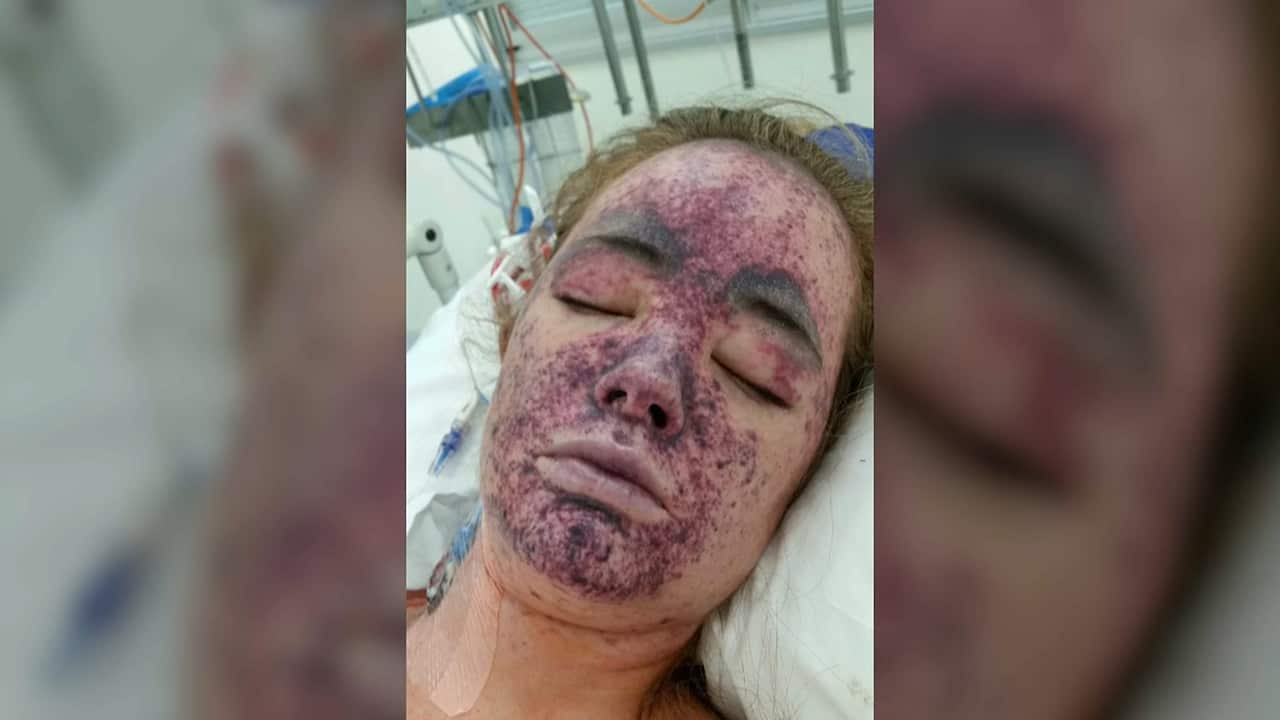
She said more needed to be done to provide vaccinations for the B strain of the infection and also to provide coverage for people aged 20 to 24.
"I do believe that there are many people who are vulnerable who aren't covered in this category and they just need to speak to their GP, and get vaccinated to stay protected," she said.
"Meningococcal is one of those things that needs a lifetime of vigilance."

More than a million teenagers are expected to get the vaccine in the next four years.
The listing for adolescents had been recommended by the Pharmaceutical Benefits Advisory Committee.
It comes after the jab became freely available for 12-month-olds in July.

Health Minister Greg Hunt said the bacterial infection, although rare, can be deadly.
It killed 28 Australians last year, compared to 11 in 2016 and 12 in 2015.
"The consequences are devastating for individuals and their families," he said.
The federal government has also been under pressure to list a vaccine for the B strain of meningococcal on the National Immunisation Program.
But the prime minister has said the change cannot occur until the vaccine earned the recommendation of an expert committee.
He has urged the company that makes the vaccine, GlaxoSmithKline, to have another attempt at becoming listed on the program.
The company confirmed in August it would consider the move but only after it had the results of a South Australian study on the impact of the vaccine, Bexsero, on meningococcal B immunity in 2019.

